 Virtually all naturally occurring cesium has 78 neutrons (Ce-133). 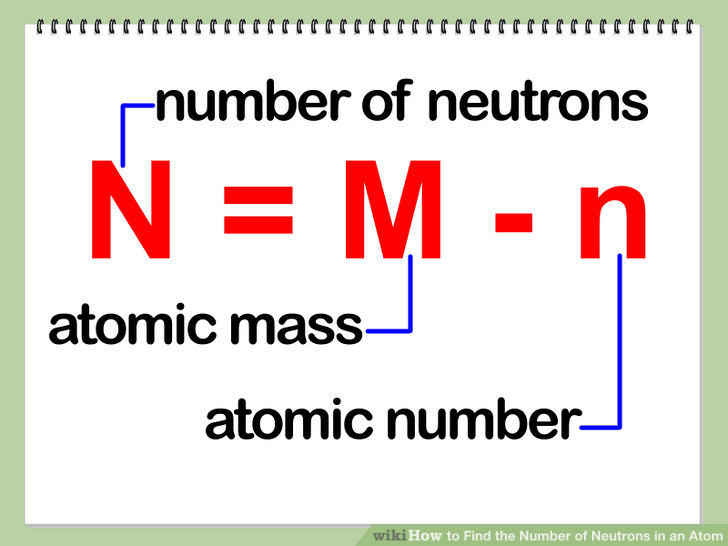
You are dealing with the calcium-40 isotope, #""_20^40"Ca"#. Cesium (caesium) has the atomic number 55 (55 protons). #"mass number" = A = "20 protons" + "20 neutrons" = 40# Frequently Asked Questions What is the atomic number of cesium The atomic number of an element refers to the number of protons (positively charged sub-atomic particles) found in a single atom of. 
Therefore, the mass number of this calcium isotope will be Calciums atomic number is 20, which means all calcium atoms have 20 protons in their nuclei. The number of neutrons can vary, but the number of protons must always be equal to #20# for a calcium isotope. Now, in order for an atom to be an atom of calcium, it Must contain #20# protons in its nucleus. the most stable isotope of cesium, Cs-133 has 78 neutrons (133 - 55 78). A quick look in the periodic table will reveal that calcium has an atomic number equal to #20#. How much protons neutrons does cesium have 55 protons are there in cesium, as its atomic number is 55. Total number of protons in the nucleus is called the atomic number of the atom and is given the symbol Z. There are 39 known isotopes and 17 nuclear isomers of tellurium, with atomic. This means that in order to get its mass number, all you need to know is how many protons are found in its nucleus.Īs you know, the number of protons an atom contains in its nucleus is given by that atom's atomic number. Summary Atomic Number Protons, Electrons and Neutrons in Caesium Caesium is a chemical element with atomic number 55 which means there are 55 protons in its nucleus. In your case, you know that an atom of calcium, #"Ca"#, has #20# neutrons in its nucleus. Cesium bromide (CsBr) Cesium chloride (CsCl) Cesium fluoride (CsF) Cesium hydride (CsH) Cesium iodide (CsI) Cesium oxide (Cs 2 O ) Cesium. Isotope: Half Life: Ca-40: Stable: Ca-41: 103000.0 years: Ca-42: Stable: Ca. Number of Neutrons: 78: Number of Electrons: 55: Melting Point: 28.5° C: Boiling Point: 678.4° C: Density: 1.837 grams per cubic centimeter: Normal Phase: Solid: Family: Alkali Metals: Period: 6. 
First thing first, you need to make sure that you have a clear understanding of what mass number actually is.Īs you know, the nucleus of an atom contains protons, which are positively charged particles, and neutrons, which are neutral particles.Īn atom's mass number will always tell you how many protons and neutrons its nucleus contains. Number of Neutrons: 20 Classification: Alkaline Earth Crystal Structure: Cubic Density 293 K: 1.55 g/cm 3 Color: Silvery Atomic Structure : Number of Energy Levels: 4 First Energy Level: 2 Second Energy Level: 8 Third Energy Level: 8 Fourth Energy Level: 2 Isotopes. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
